Like solids, liquids and gases are also capable of heat transfer by thermal conduction. But the extend of conduction is less compared to solids. This tutorial discussing about how conduction heat transfer carried out in liquids and gases with the governing formulas.
We know that gap between molecules is higher in liquids and gases than closely packed structures usually found for solids. Since conduction is occurring by the direct interaction between molecules by vibration and hence transferring kinetic energy during molecular impact. Since molecules are much apart from each other in liquids and gases, therefore the amount of heat transfer by conduction is also less in liquids and gases when comparing to the high heat conduction in solids.
[h=Conduction in liquids]2[/h]
In liquids, conduction heat transfer is small, since random translatory motion is small. Transfer of energy in liquids is occurred by longitudinal vibration similar to the propagation of sound. To know more see modes of heat transfer
It is worthwhile to consider Bridge man assumptions for conduction heat transfer in liquids. They are
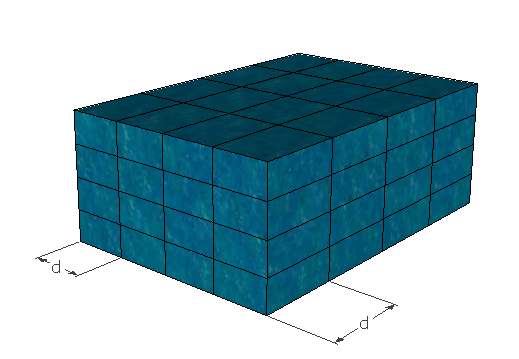
1. In liquids, adjacent molecules are arranged in simple cubic fashion as shown
2. Transfer of energy from one molecule to another is occurring by longitudinal vibration, with the velocity of sound.
From these assumptions, we can found the expression for thermal conductivity in liquids
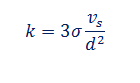
Where d is the cell dimension as shown, s is the Boltzmann's constant/molecules. v_s is the sonic velocity.
Conductivity of liquids can also be explained in terms dynamic viscosity µ as
Where 'n' is the number of atoms in a molecule.
[h=Conduction in gases]2[/h]
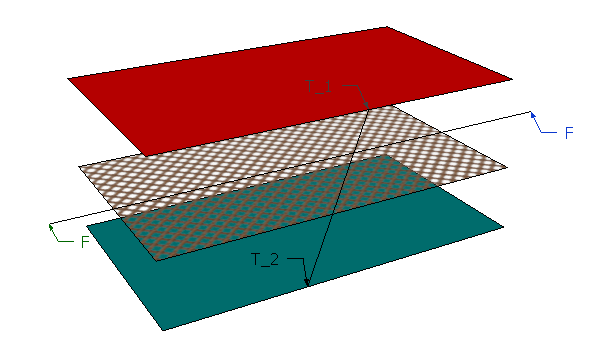
Consider we have an imaginary surface F-F in a gas have temperature difference of T_2-T_1 between two layers of surface. Then molecules at higher temperature surface will have higher velocity of vibration than that in the lower temperature region. Hence there is a possibility of conduction heat transfer through this imaginary surface.
Thermal conductivity of gases can be found out using the relation
Where
n is the number of molecules per unit volume, f is the number of degrees of freedom, d is the molecular mean depth path, s Boltzmann's constant and v_s men molecular velocity.
From the above relation, thermal conductivity is proportional to mean free path. Since molecular free path of gas molecules decreases with increase of density, hence molecule with higher molecular weight will have lower thermal conductivity than lower molecular weight gases. Just take an example of comparison of thermal conductivity between hydrogen and nitrogen gases.
K for nitrogen is 0.021 W/mk which is lower than lower molecular weight gas hydrogen having thermal conductivity of 0.15 W/mk at standard temperature and pressure.





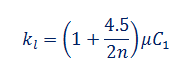
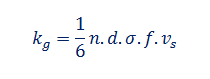

 Reply With Quote
Reply With Quote

Bookmarks