Conduction is the method of heat transfer usually we experience in our day to day activities. Thermal conduction is carried out by either molecular vibration or by movement of free electrons. There is no molecular movement in conduction heat transfer. This tutorial narrates thermal conduction in detail with the help of Fourier’s law of thermal conduction.
We learned that conduction heat transfer occurs between two bodies which are in thermal contact or between two parts of the same body having temperature difference or temperature gradient between them see modes of heat transfer. Conduction heat transfer is found to be occurring commonly in solids in which the molecular movement is restricted when comparing with a liquid or gas. Conduction heat transfer in liquids and gases are found to a lesser extent. See: Thermal conduction in liquids and gases.
In solids as temperature increases molecules will begin to vibrate. Molecules having higher temperature at the hot part of the body will vibrate with more agitation than molecules at the lower temperature part of the body. In conduction heat transfer kinetic energy is translated between the molecules which are very adjacent to each other while vibrating. And this energy is kept on transferring from hot region to the cold region or from hot body to the cold body. Since the gap between the molecules in liquids and gases are high, hence conduction heat transfer by vibration interaction is quite low in liquids and gases.
In metals, conduction occurs mainly by the movement of free electrons inside the metal atoms. This is the reason for why good electric conductors are also good thermal conductors. example: Silver, Copper. Free electrons provide an energy flux in the direction of decreasing temperature.
The ability of a material to conduct heat is depends upon its value of thermal conductivity K. Material having higher values of thermal conductivity are good conductors and with low value of thermal conductivity are good insulators. Sequences of materials in decreasing order of thermal conductivity are
1. Pure metals
2. Metal alloys
3. Non-metallic crystalline and amorphous substances
4. Liquids
5. Gases
[h=Fourier's law of thermal conduction]2[/h]
Detailed study on conduction was performed by French scientist Fourier. He derived the basic empirical formula based on observations and assumptions which is taken as the datum of conduction heat transfer.
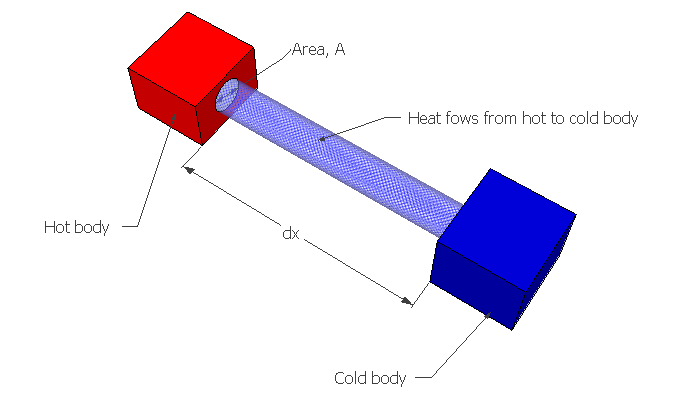
According to Fourier's law of heat conduction, the rate of heat flow is directly proportional to the area of the section at right angles to the flow of heat, and also to the change in temperature over the length of heat flow.
Mathematically,
Where Q is the rate of heat flow, A is the area perpendicular to the flow, 'dt' is the temperature difference between length 'dx' and k is thermal conductivity of the substance as I already mentioned before. –ve sign indicates that decreasing temperature along increasing the length or thickness. To know more see one dimensional heat conduction.



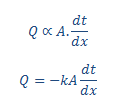

 Reply With Quote
Reply With Quote

Bookmarks